MEET THE CORE: SUSAN BRENNER-MORTON
Susan Brenner-Morton is an antibody expert who has spent decades developing these specialized tools to help us understand how motor neurons function normally—and dysfunction in ALS. We sat down virtually with Susan, who is Director of the Antibody Unit of the Project ALS Therapeutics Core at Columbia and Director of Custom Antibody Services and Senior Science Officer at Columbia’s Zuckerman Institute for Mind, Brain, and Behavior, for a look at her background, her current research focus, and why a custom antibody unit is essential to drug assessment and development at THE CORE.
What exactly is an antibody?
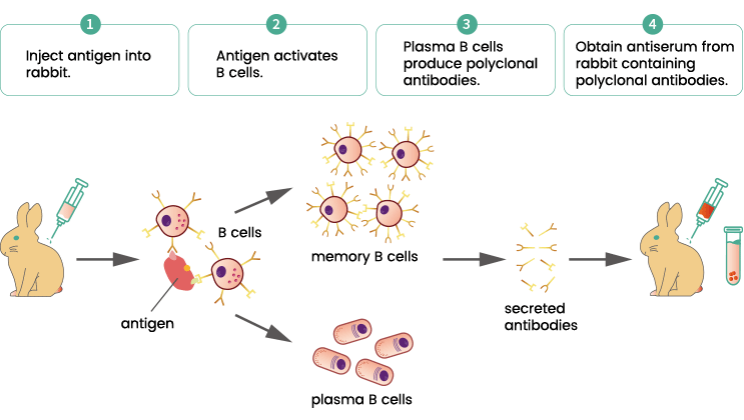
Antibodies are protein molecules that are made by B cells in response to the body being invaded by some infecting organism or virus. When things are working properly, your body shouldn’t be making antibodies to your own proteins. Each B cell makes one unique antibody that targets one epitope, or specific piece of the invading protein.
To make an antibody in the lab, you immunize an animal with the protein you want to generate antibodies to, which activates the specific B cells that express a receptor for that piece of protein. This causes the B cells to divide and generate both memory cells and plasma cells which secrete antibody. The next time the animal sees the immunizing protein those memory cells are activated. With repeated immunizations, the antibody response gets stronger and continues to become a better fit to the protein, leading to a higher antibody titer—more and better antibody.
How did you come to develop novel antibodies for brain- and specifically motor neuron-focused research?
My Masters is in microbiology/immunology and my initial work experience was centered around diagnostic bacteriology. I first learned to make monoclonal antibodies after moving to Seattle where I worked in a tumor immunology lab. In 1986, I moved back to New York and joined Tom Jessell’s newly formed lab, which had not yet established a clear direction (although he was always interested in motor circuitry as the basis for all behavior. As people with ALS learn, without the ability to move, there is no means of output—you are a prisoner with only your thoughts). In our early experiments, we took an unbiased approach: I made antibodies to cell membranes of whole spinal cord and looked for interesting staining patterns on tissue sections. I had a naive eye and unlike Tom, I had no expectations of what I wanted to see. Anything that lit up an interesting pattern was worth saving. Antibody staining is very visual and led to some very important lines of inquiry. Some discoveries were made by accident, and understanding their importance required rigorous and disciplined study. As the investigative path of the lab developed, we started targeting specific structures and then specific molecules with antibodies to assess complex questions around how neurons develop and function, normally and in disease states like ALS.
Why is it important for researchers in THE CORE to be able to access custom antibodies? What are a few of the antibodies that you have made or are working on in THE CORE?
Antibodies, when purchased from a company, are extremely expensive. They also can vary in their quality from lot to lot. It is important to have reproducibility. When you make your own reagent, you can target certain epitopes to ensure specificity. A successful project can provide you with enough working antibody to last for years. For some molecules, good antibodies don’t exist.
Some of the antibodies I have made and am working on currently are not ALS specific antibodies but define structures or cell types involved in motor neuron circuitry. One such antibody is directed against synaptophysin, which is present when motor neurons form synapses with skeletal muscle and can therefore be used to evaluate denervation of the muscle. Another such antibody I am currently working on is a choline acetyltransferase (chat) antibody. Chat antibodies label motor neuron cell bodies and can directly indicate the amount of motor neuron cell loss when assessing an experimental model.
I am also making new antibodies directed against proteins directly implicated in ALS pathology. Mutant FUS proteins that can cause ALS, for example, are often undetectable using existing FUS antibodies that were not developed with the ALS-relevant mutations in mind. I have developed several antibodies against specific ALS FUS mutations that THE CORE can now use in the research lab and the clinic to more accurately assess disease progression and potential therapeutic benefits. I am also working on monoclonal antibodies against TDP43 and C9orf72 Di-Peptide Repeats (DPRs) GR and PR. All antibodies can be used diagnostically, and monoclonal antibodies can also be humanized for potential use as therapies.
What are the most important criteria when considering whether an antibody could be used as a therapy?
The first criteria for developing a therapeutic antibody is that it is capable of binding to the protein in its native conformation – i.e., inside a person’s body. Importantly, the antibody should have a high affinity for its target, and be able to block the function/pathway of the target molecule by either blocking the functional domain of the target or by clearing it from the body and making it inaccessible. Overall, a therapeutic antibody must be humanized to avoid causing an immune response in people, it must be very specific, and for neurological diseases, it must be able to cross the blood brain barrier to reach its target.
Stay tuned for a live interview with Susan on a future Project ALS webinar, where she will answer specific questions about different types of antibodies and challenges in making specific and reproducible antibodies, and progress in antibody development.

Stories of the Season: The Harper Family
One Day at a Time: Jacob Harper’s Journey With ALS Jacob Harper is 26 years old, from southern West Virginia, and living with ALS. According


